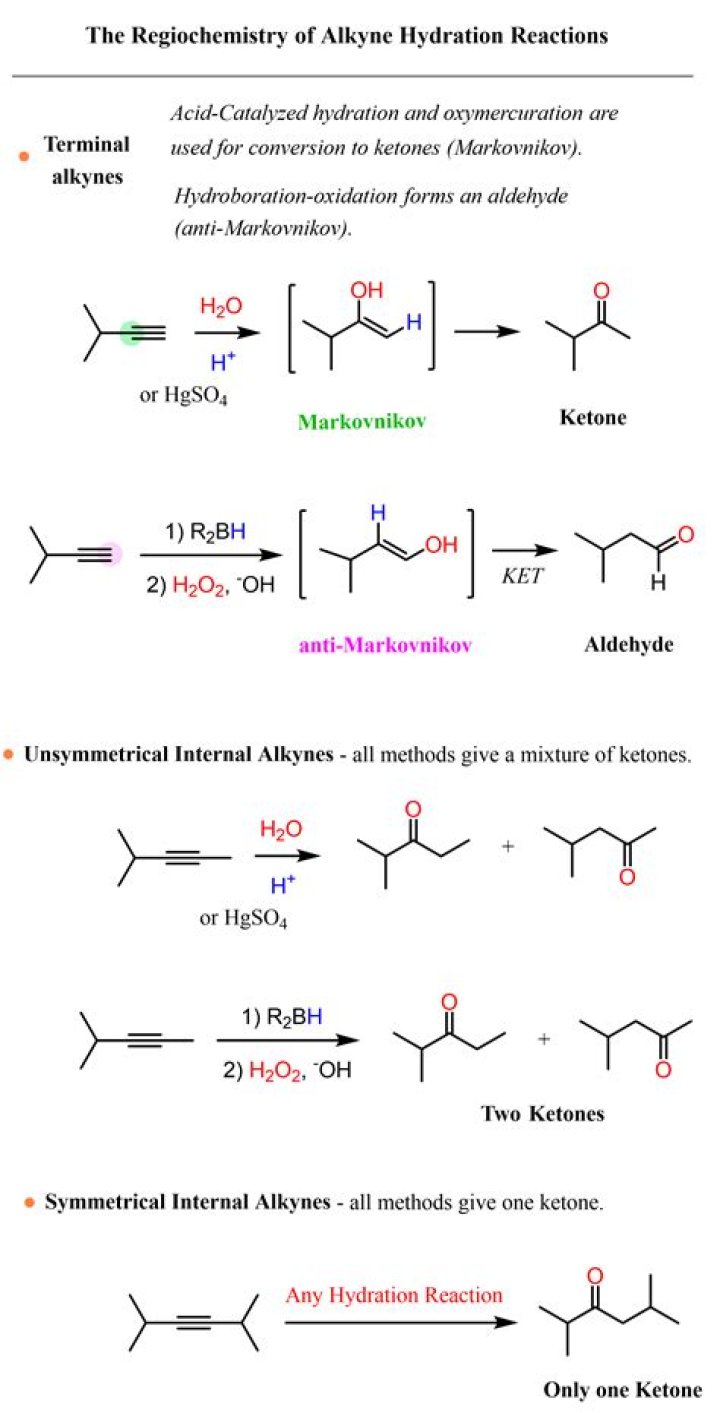
The mercury(II) catalyzed hydration of a terminal alkyne produces a methyl ketone, while the hydroboration-oxidation produces an aldehyde. For internal alkynes, the regioslectivity of these reactions are rendered ineffective. The reactions are redundant in that they both produce the same ketone products.
Which reagent can hydrate an alkyne?
As with alkenes,hydration (addition of water) to alkynes requires a strong acid, usually sulfuric acid, and is facilitated by mercuric sulfate.
Which alkyne gives aldehyde on hydration?
acetaldehyde
The hydration of acetylene indeed gives only acetaldehyde. (In fact, acetaldehyde is the only aldehyde that can be prepared by the hydration of an alkyne.)
What is the hydration of alkynes?
Hydration of alkynes begins similar to the hydration of alkenes through the addition of the first water molecule. As with alkenes, hydration (addition of water) to alkynes requires a strong acid, usually sulfuric acid with a mercuric sulfate catalyst as shown below.
Do alkynes react with water?
Water addition to terminal alkynes leads to the generation of aldehydes, while nonterminal alkynes and water generate ketones. Water adds across the triple bond of an alkyne via a carbocation mechanism. Dilute mineral acid and mercury(II) ions are needed for the reaction to occur.
What is hydration alkynes?
Is hydration of alkynes markovnikov?
Just as alkenes, alkynes can be hydrated by two different methods. The direct addition of water catalyzed by mercury(II) salts yields the Markovnikov product. In contrast, the indirect hydration by the reaction sequence of hydroboration, oxidation and hydrolysis results in the anti-Markovnikov product.
Do alkynes undergo hydration?
With the addition of water, alkynes can be hydrated to form enols that spontaneously tautomerize to ketones. The reaction is catalyzed by mercury ions and follows Markovnikov’s Rule A useful functional group conversion for multiple -step syntheses is to hydrate terminal alkynes to produce methyl ketones.
What happens when water is added to alkynes?
Hydration. The addition of the elements of water across the triple bond of an alkyne leads to the formation of aldehydes and ketones. Water addition to terminal alkynes leads to the generation of aldehydes, while nonterminal alkynes and water generate ketones.
Why do we use HgSO4 as a catalyst in alkene hydration?
In alkene hydration, only dilute H 2 SO 4 is used. But due to slow reaction rate of alkynes and H 2 SO 4, we have to use a catalyst. Therefore we use HgSO 4 as the catalyst to increase the reaction rate. Products of alkyne with HgSO 4 and H 2 SO 4 reaction
How do you hydrate alkynes?
Just as alkenes, alkynes can be hydrated by two different methods. The direct addition of water catalyzed by mercury (II) salts yields the Markovnikov product. In contrast, the indirect hydration by the reaction sequence of hydroboration, oxidation and hydrolysis results in the anti-Markovnikov product.
What is the difference between Alkyne hydration and ethyne hydration?
HgSO 4 and aqueous dilute H 2 SO 4 is used for alkyne hydration. Alkynes hydration is much harder than alkene hydration. Carbonyl compounds are given as products. Carbonyl compounds are grouped as aldehydes are ketones . Only ethyne hydration will give an aldehyde compound, ethanal. Ethanal is the simplest aldehyde.
What is the mechanism of hydration of alkene?
Acid-Catalyzed Hydration Mechanism 1 The pi electrons of the alkene attack a hydrogen of H3O+ resulting in carbocation formation. 2 When there is no favorable rearrangement water carries out nucleophilic attack on the carbocation forming an oxonium ion. 3 A water molecule deprotonates the oxonium intermediate yielding the product alcohol.