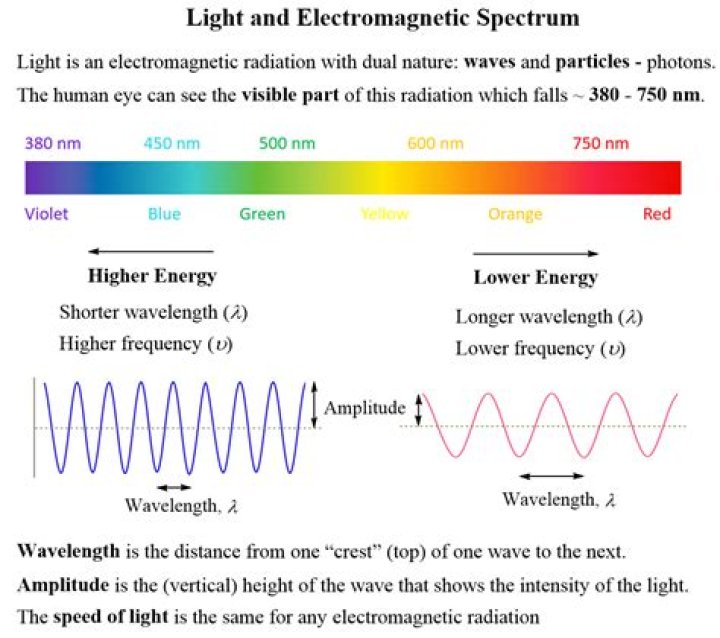
The amount of energy is directly proportional to the photon’s electromagnetic frequency and thus, equivalently, is inversely proportional to the wavelength. The higher the photon’s frequency, the higher its energy. Equivalently, the longer the photon’s wavelength, the lower its energy.
How do photons relate to the electromagnetic spectrum?
Electromagnetic radiation can be described in terms of a stream of mass-less particles, called photons, each traveling in a wave-like pattern at the speed of light. Each photon contains a certain amount of energy. The different types of radiation are defined by the the amount of energy found in the photons.
Does wavelength affect number of photons?
Your premise about the number of photons and energy is not quite correct. They are related by λ = hc/ E photon where h is Plank’s constant, c is the velocity of light and λ is the wavelength of the photon. In a given packet of photons the total energy is proportional to the number of photons.
Do photons have a wavelength?
Generally speaking, photons have similar properties to electromagnetic waves. Each photon has a wavelength and a frequency. The wavelength is defined as the distance between two peaks of the electric field with the same vector. Unlike an electromagnetic wave, a photon cannot actually be of a color.
What relationship exists between wavelength and frequency between frequency and energy between wavelength and energy?
Just as wavelength and frequency are related to light, they are also related to energy. The shorter the wavelengths and higher the frequency corresponds with greater energy. So the longer the wavelengths and lower the frequency results in lower energy.
What is the relationship between wavelength frequency and energy of electromagnetic waves?
Just as wavelength and frequency are related to light, they are also related to energy. The shorter the wavelengths and higher the frequency corresponds with greater energy. So the longer the wavelengths and lower the frequency results in lower energy. The energy equation is E = hν.
What does wavelength and frequency have to do with the EM spectrum?
Electromagnetic waves are categorized according to their frequency f or, equivalently, according to their wavelength λ = c/f. Visible light has a wavelength range from ~400 nm to ~700 nm. Electromagnetic waves with shorter wavelengths and higher frequencies include ultraviolet light, X-rays, and gamma rays.
What is the relationship between wavelength velocity and frequency?
When the wavelength of the wave increases and the frequency is constant, the velocity of the wave also increases because the wavelength is directly proportional to velocity. When the frequency of a wave increases and the wavelength is constant, the velocity of the wave increases.
What is the relationship between wavelength and frequency in electromagnetic waves?
Section Summary. The relationship among the speed of propagation, wavelength, and frequency for any wave is given by vW = fλ, so that for electromagnetic waves, c = fλ, where f is the frequency, λ is the wavelength, and c is the speed of light.
How does wavelength affect the photoelectric effect?
In the photoelectric effect light which strikes a metal causes electrons to be emitted. As the wavelength decreases for a specified metal, the speed (and thus the Kinetic Energy) of the emitted electrons increases.
How do photons have a wavelength?
The photon is an elementary particle in the standard model of particle physics. It does not have a wavelength. It is characterized in the table as a point particle with mass zero and spin one.
What is the relationship between photon energy and wavelength?
Photon Energy and Wavelength. The energy of light (or photon energy), E, is inversely proportional to the wavelength by the equation: E = hc/ l (equation 1) where h is Planck’s constant (6.6261 x 10 -34 J*s) and c is the speed of light (2.9979 x 10 8 m/s).
How do you calculate the energy of a photon?
It is calculated using either the transverse frequency or wavelength equations (frequency is wave speed divided by wavelength). These equations can be used for any atom using the Photon Frequency or Photon Wavelength equations. Photons beyond hydrogen are often reported in terms of energy, not frequency or wavelength.
How are the photon wavelengths for hydrogen accurately calculated?
The photon wavelengths for hydrogen are accurately calculated using this equation. In hydrogen photon wavelength calculations, the wavelengths of absorbed photons for hydrogen were calculated at differing orbitals when the atom is ionized.
What is the difference between high energy photons and low energy photons?
Low energy photons (such as radio photons) behave more like waves, while higher energy photons (such as X-rays) behave more like particles. The electromagnetic spectrum can be expressed in terms of energy, wavelength or frequency.